I spoke with Tony Larkman, CEO of the Australian Tea Tree Industry Association, about the nonprofit he heads, his own introduction to essential oils, and issues facing the essential oils industry and tea tree oil in particular.
Tony Larkman was born and raised in Kenya. After travelling extensively through Africa, Europe and the Middle East he returned to East Africa in 1986 to work first as a purchasing officer and then as a pig farmer before moving to Australia in 1992. Over the next 17 years Tony turned his hand to feed milling and grain trading before becoming an IT Manager and then a Purchasing Manager. In 2009 he started working for ATTIA Ltd where he realized that he had finally stumbled on the answer to a question that had plagued him since he was child: “What do you want to be when you grow up?” Tony is now the CEO of ATTIA Ltd which is probably the most organized and active industry body for any single essential oil.
RT: Your introduction to tea tree oil was as a pig farmer?
TL: It was the mid-1980s, and a spray bottle of ‘healing oil’ with tea tree oil as the active ingredient was hung near the door of every building on the piggery. We used it everywhere and for everything, for both workers and animals – from minor cuts and scratches through to cleaning and preparing wounds for stitching after fights between boars – some of which were horrendous and likely to have been fatal without the magic of the healing oil. It also helped mask some of the smell of a piggery, which was a bonus.
What is ATTIA, and what services does it provide?
The Australian Tea Tree Industry Association (ATTIA Ltd) is an Australian based not-for-profit organization formed in 1986 as the peak body to promote and represent the interests of the Australian tea tree industry. From the grower/producer to the manufacturer of off-the-shelf products for public use, ATTIA supports and promotes the responsible use of pure Australian tea tree oil (TTO). ATTIA’s aim is to develop a stable, cohesive, environmentally friendly, and internationally competitive TTO industry producing quality assured TTO that meets or exceeds international standards. ATTIA promotes the safe effective use of TTO for a wide range of applications. More information about ATTIA here.
Full membership of ATTIA is open to all Australian tea tree industry participants while associate membership is open to international participants in the supply chain. Our services include:
- Support and advocacy in dealing with government and regulatory agencies, research and development groups and the media.
- Leadership and representation in the formulation of overall industry strategy.
- Generic industry promotion at national and international levels.
- The collection and compilation of market data to inform all links in the supply chain of factors influencing supply and demand for pure Australian TTO.
Is the ATTIA website useful for consumers, enthusiasts or practitioners?
ATTIA’s website was created to cater to all levels of interest from school children preparing a project through casual consumers and passionate enthusiasts to professionals. The most commonly accessed pages which are easily navigated to by using the drop-down boxes in the navigation pane at the top of every page are:
Home Page A summary of who ATTIA is and where pure Australian tea tree oil comes from.
About Tea Tree A more in-depth look at production, uses, safety and efficacy.
FAQ Answers 20 of the most commonly asked questions on tea tree oil.
For professionals and researchers there is also a regularly updated, searchable Literature Database where detail on more than 1,200 papers can be accessed. These include most articles published on the safety, efficacy and uses of pure tea tree oil since 1904 when the species from which pure tea tree oil is distilled (Melaleuca alternifolia) was first described by Maiden & Betche.
How much tea tree oil is produced annually in Australia and around the world and to where is it distributed?
We know that between 400 and 500 metric tons of pure Australian TTO is produced annually; this is dependent on the weather and growing conditions from year to year. 80% of this is exported, most to North America. In 2012 a total of 409 metric tons of Australian TTO was exported to the following destinations:
- 50.27% North America (USA, Canada, Mexico)
- 25.66% Europe (Continental Europe, UK, Russia)
- 22.76% Asia (India, Middle East, South East Asia, China, Japan)
- 0.98% Africa (South Africa, Egypt)
- 0.33% South America (Brazil, Chile, Colombia)
Tea tree oil is also produced in several other countries including China, South Africa, Kenya, Indonesia and Thailand. All of this TTO is produced from Melaleuca alternifolia, an Australian native tree that originated in the coastal regions of Southern Queensland and Northern NSW. No one can accurately state how much TTO is produced in these other countries.
Are there different qualities of tea tree oil?
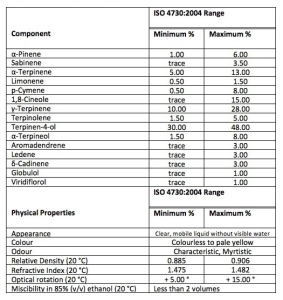
Let me clarify first that there is no ‘pharmaceutical grade’ of TTO, nor is there a ‘therapeutic grade’. These are purely marketing inventions intended to convince a buyer that a particular brand is in some way superior to that of competitors. The main ingredient and most active bacteriostat in TTO is terpinen-4-ol which, according to the current standard, must be between 30% and 48% in TTO. Almost all pure Australian TTO is traded today with 40%+ terpinen-4-ol. The only way to make sure you are getting the best available TTO is to ask the retailer or manufacturer if they are using pure Australian TTO and how they verify this. If they can’t (or won’t) answer then either they are cheating or they genuinely don’t know. If the latter is true please refer them to the ATTIA website or Facebook page.
The second great marketing myth that has been roundly debunked in scientific literature is the belief that the level of another compound invariably found in pure Australian TTO: 1,8-cineole, needs to be as low as possible to prevent irritation. Today’s standard allows up to 15% of 1,8-cineole (which is actually the main constituent of another Australian native essential oil – eucalyptus oil – which contains around 80% of 1,8-cineole, or eucalyptol as it is also known) and there is clear evidence that it makes no difference in TTO at levels of up to 8%.
Tea tree oil is steam distilled from the leaves and terminal branches of the Australian native tree Melaleuca alternifolia. When correctly distilled, stored and shipped any oil extracted from M. alternifolia is just as safe and efficacious whether it is produced in Australia or anywhere else in the world. In Australia all production, distillation, storage and shipment processes are stringently controlled through the ATTIA Code of Practice (COP) quality assurance program to guarantee that only pure, natural TTO derived from M. alternifolia is supplied in perfect condition to discerning manufacturers both locally and around the world. In other countries issues related to pesticide use, identification of source plants, poor distillation, as well as poor handling and storage practices can contaminate or degrade the oil to the point where it is no longer safe to use and cannot even be described as tea tree oil.
“TTO has an enviable reputation for its
antibacterial, antifungal, anti-inflammatory,
anticancer and antiviral properties”
In addition to these quality assurance issues, as TTO passes up the supply chain, some unscrupulous suppliers and traders deliberately contaminate it with byproducts that are derived either from other essential oils such as pine oil or from synthetic production processes. This adulteration can significantly compromise the safety and efficacy of the product when used as a replacement for pure TTO. In some instances I believe this practice has led to TTO being abandoned as a traditional herbal product because either ‘it doesn’t work’ or ‘it causes irritation’. The most frustrating part of this for ATTIA is that the claims made by those who sell the adulterated material are based in the main on ATTIA sponsored science into the safety, efficacy and uses of pure Australian TTO. More information on adulteration is available from a Facebook site, which was created specifically to raise awareness of adulteration in TTO.
These days pure Australian TTO is traded in commercial quantities using the International Standards Organization’s standard ISO 4730:2004 but the oil typically has 40% or more terpinen-4-ol, and a maximum of 3-4% 1,8-cineole, depending on the buyer. Occasionally you can still see “T39-C2”,”T39-C4” or “T40-C4” on some bottles; all they are doing is trying to differentiate their product from that of their competitors. The fact remains that there is clear scientific evidence that pure TTO is just as safe and effective regardless of the levels of terpinen-4-ol or 1,8-cineole as long as it conforms to ISO 4730:2004 standards. More information on the ISO standard for pure Australian TTO can be found here.
What are the principal issues that impact tea tree oil and essential oil quality?
I have already touched on adulteration as a major issue and I believe this is the most significant quality threat we currently face as an industry. However, even pure TTO can be degraded over time due to poor distillation, storage and handling so producers, traders, manufacturers and end users need to be aware of these factors and minimize their effect. Exposure to light, high temperatures and atmospheric oxygen all lead to degradation of some of the components of TTO which can increase the incidence of skin irritation and sensitization.
On the ATTIA website in the section about packaging you can find a paper on the stability of pure Australian TTO which can also be downloaded here. This details the science behind storage and handling procedures. A quick summary:
- ATTIA recommends that pure Australian TTO sold to the public should only be stored in small (up to 100 ml) dark glass bottles at temperatures not exceeding 25oC with a use by date of 2 years from the date of manufacture in unopened bottles.
- Once opened these bottles should be stored in cool, dark conditions with the cap tightly sealed and discarded after 6 months if still unused.
- For bulk storage, the ATTIA Code of Practice requires producers to use only stainless steel storage and transport vessels and to store the oil in cool, dark conditions with minimum exposure to air.
- ATTIA recommends 3 years from filling as the use-by date for pure Australian TTO sold in correctly filled, purged (Nitrogen or Argon) and tightly sealed stainless steel drums stored at an ambient temperature not exceeding 25oC.
What do you see as the outstanding benefits of tea tree oil?
TTO has an enviable reputation for its antibacterial, antifungal, anti-inflammatory, anticancer and antiviral properties, which are all well supported scientifically. In 2013, TTO was exported from Australia to 43 countries, where its uses include cosmetic, industrial, and medicinal applications. It is interesting occasionally to see that in advertising for some new or emerging essential oils, comparison is most often made to the efficacy and safety of Australian TTO – reinforcing its position as a global leader.
Australian TTO is a natural product that contains more than 113 compounds and I believe that it is the synergistic effect of these compounds that makes it a true healing oil. Some components like terpinen-4-ol (the major constituent of TTO) have proven antimicrobial and antifungal properties on their own but there are all the other components which must assist and complement the terpinen-4-ol with factors such as skin penetration, bactericidal, fungicidal and anti-inflammatory activity.
Another outstanding benefit is its proven ability to control commonly occurring bacteria such as MRSA and VRSA that are rapidly becoming totally resistant to all conventional antibiotics, particularly in hospital environments where management of these infections is proving such a challenge to healthcare professionals. I believe that as we run out of control options, more and more health professionals will turn to products like TTO that have not only proven antimicrobial activity but also possess a natural complexity that makes development of bacterial or fungal resistance extremely unlikely.
What current or upcoming research projects is ATTIA involved in?
Over the last 20 years the Australian tea tree industry has been fortunate to enjoy a close partnership with the Australian Government’s Rural Industries Research and Development Corporation (RIRDC). RIRDC has co-funded much of the research, development and extension work done to develop our industry through a tree breeding program, agronomy advances, development of distillation techniques and safe storage and handling procedures for pure Australian TTO. In addition to these basic but vital tools RIRDC has co-funded much of the University based research into the safety, efficacy and uses of TTO resulting in more than 50 publications which are available for download from the TTO page on the RIRDC website here.
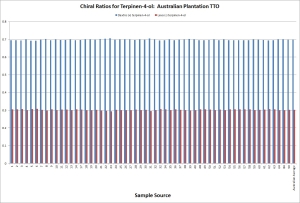
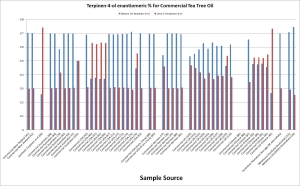
Currently ATTIA is developing a test for adulterated oil which we hope to have adopted by internationally recognized standards organizations in the near future. Called the Chiral Purity Test, this measures the ratios of the optical isomers of three compounds that occur in pure Australian TTO: terpinen-4-ol, α-terpineol and limonene. Each one of these compounds occurs in two forms, with specific ratios in TTO. Since we know what these ratios should be, any variation is a strong indicator that the product has been contaminated. Table 1 below illustrates 57 samples of Australian TTO sourced directly from plantations over five years (2007 – 2011) showing a consistent ratio (70:30) of the two terpinen-4-ol enantiomers (dextro and levo). Table 2 shows 47 samples of commercially available TTO sourced from around the globe as well as a sample of 98% pure terpinen-4-ol. Some have a significant deviation from the expected ratio for pure TTO, indicating adulteration. This demonstrates how easy it is to identify real TTO from adulterated material. As well as synthetic terpinen-4-ol, potential contaminants include industrial waste from ‘normalising’ pine, eucalyptus and other essential oils. ATTIA intends to use the test to “out” any offenders by reporting them to the relevant regulatory bodies in their jurisdictions.
ATTIA is also keenly following the progress of researchers at the University of Western Australia (UWA) who are investigating the anti-cancer potential of pure Australian TTO. A few years ago topical application of a very specific formulation containing pure Australian TTO as the active ingredient was shown to significantly reduce the viability of melanoma and mesothelioma cancer cells in vitro and since then studies of skin cancers in mice have confirmed its ability to reduce or resolve some types of skin cancers. Researchers believe this may be due to the stimulatory effect TTO has on the body’s own immune system. Much work remains to be done before this reaches proof-of-concept stage.
Researchers at UWA are also investigating the clinical activity of formulated products containing TTO for activity against acne-causing bacteria. This will eventually result in a pilot study using volunteers to measure the activity of the most promising formulations in a human population.
For the grower members of ATTIA there is continuing support for research into high yielding genotypes of M alternifolia through both a conventional breeding program and using cutting-edge technology to identify specific gene markers for high yield and disease resistance that occur in natural populations. These markers can be used to quickly and efficiently identify individuals with outstanding properties from existing wild populations for infusion into the ongoing conventional breeding program to increase genetic diversity and ensure sustainability. In an emerging area ATTIA and the Australian Government have recently commenced a joint study to investigate the use of biochar generated from spent leaf and inter-crop legume planting to reduce both carbon and nitrous oxide emissions. Increasing productivity and developing a low emission industry not only ensures survival of an industry that utilizes a native crop plant in a sustainable way but also improves the green credentials of an already clean and green industry.
Tea tree oil safety issues that have been raised in the past include skin sensitization, endocrine disruption and bacterial resistance. What is your take on these?
I want to address these separately as all three are critically important questions and need different answers.
Allergic reactions and skin sensitization
TTO that has been well stored since distillation is a safe, natural product and its efficacy and safety is well known and documented. There is absolutely no doubt that some people are allergic to pure Australian TTO in the same way that some people are allergic to peanuts, milk or wheat. This can manifest itself in many forms from reddening, irritation and itching of the skin through to blisters and burns. Whenever and wherever TTO is used for the first time a user should make sure that it is diluted in a carrier oil (eg jojoba) to 10% before trying a small drop on the soft skin of the inside of the forearm. Wait 24 hours, unless immediate signs of itching or swelling are noticed. If a reaction occurs, immediately wash the area with soap and water and discontinue use. It is worth mentioning that slight, transient irritation is sometimes seen in clinical trials, this may be considered acceptable, depending on the benefits. However, allergic reactions are never acceptable.
Immediate, strong allergic reactions to TTO are uncommon and have been estimated to occur in less than 0.5% of the population. Sensitization to TTO has also been well documented; this occurs where repeated exposure to a product increases the reaction to its application. This can happen with pure TTO, but it is more likely to occur where TTO has either been poorly stored, which increases the level of peroxides and other irritants through oxidation, or when the oil has been adulterated.
Endocrine disruption
This is a myth and is the one that get me the most upset. I often see reference in scientific articles, blogs and online comments that refer to a link between gynecomastia (man boobs) and lavender and tea tree oils and I have been battling the negative publicity that erupted when the original work by Henley et al http://www.ncbi.nlm.nih.gov/pubmed/17267908 was first published in the NEJM in 2007. The authors linked the use of personal care products containing lavender and/or tea tree oil to breast development in 3 prepubertal boys and then went on to “demonstrate” that the endocrine disruption was caused by the two essential oils using well trays in the laboratory. These 96 well trays, made of plastic, contain phthalates which are known endocrine disruptors and I know from personal experience that TTO is an excellent solvent and extracts phthalates from any unprotected plastic it comes in contact with very quickly and efficiently. I believe this is why Henley et al reported the estrogenic and anti-androgenic “activity” of TTO and lavender possibly without even realizing the truth. More here.
If you look at this another way, at least 400,000 kg of pure Australian TTO is shipped annually and used by thousands of people in hundreds (or even thousands) of products as well as being applied as an undiluted oil for medical, cosmetic and aromatherapy uses every day. If TTO did indeed have any kind of estrogenic or other endocrine disruptive activity then surely gynecomastia or at least some other symptoms would have been seen in more than the three boys Henley et al looked at.
Bacterial resistance
The suggestion that exposure to sub-lethal levels of TTO induced resistance in bacteria was first reported in 2007 and again in 2008 by McMahon et al. In 2008, and again in subsequent years, researchers at the UWA TTO research group tried to replicate the work done by McMahon et al to no avail. Repeated attempts by scientists at the University of Western Australia (Hammer et al 2008, Hammer et al 2012, Thomsen et al 2013) failed to induce resistance to any antibiotics in any of the bacteria studied and led them to conclude in 2013 that “…there is no evidence to suggest that tea tree oil induces resistance to antimicrobial agents.” Unlike most antibiotics, which are usually single molecules, TTO contains more than 113 naturally-occurring molecules. The synergistic effect of these compounds makes it improbable that bacteria could develop resistance to TTO.
Do you feel that current legislation impacting tea tree oil is reasonable, and do you anticipate more restrictive legislation?
We live in a world where regulation plays a major part in the registration and use of any cosmetic or medicinal ingredient whether natural or synthetic in origin. Regulations are there to protect users from harm. Some products and chemicals are very harmful if used incorrectly or indiscriminately so we all have to accept that regulation is a fact of life; ATTIA’s approach is and always will be cooperative. Having said that it is worth pointing out that legislation varies far too much around the world and I would prefer a more uniform approach to this in the same way that many countries are now adopting and moving towards the UN’s Globally Harmonized System of Classification and Labeling of Chemicals (GHS). This makes legislation far easier to manage.
Restrictive legislation is frustrating to any essential oil industry simply because most of it is designed to work with a single molecule which is clear-cut, easy to identify and assay, and is usually also patentable. An essential oil is a variable complex of molecules that, like the human genome, belongs to all of us and at the same time to none of us – in other words a regulator’s nightmare! However there is also no doubt that these oils are efficacious, safe and widely used around the world in cosmetic and medicinal formulations so I believe that regulators need to look closely at these naturally occurring substances and create separate legislation for them. This is beginning to occur in some places, but while these are being developed essential oil industries are being forced to address two separate sets of legislation, which can be burdensome.
What are the principal challenges facing the tea tree oil industry?
In the short to medium term adulteration of a pure, natural product with byproducts and other waste from chemical factories is the foremost challenge facing our industry today. This practice is motivated purely by profit and relies totally on the safety, efficacy and reputation of pure Australian TTO while undercutting the market price often to the point where a farmer cannot safely and sustainably produce the original product. The result of this cheating is to devalue TTO as a viable product. Unless it is combated vigorously, this could lead to adulterated or wholly synthetic concoctions becoming the norm for customers buying or trading ‘tea tree oil’ resulting in a devalued and virtually useless substitute for the real thing. ATTIA is currently cooperating with scientists and regulators from around the world to develop a cheap, effective test that will allow a discerning manufacturer to detect some of the more common forms of adulteration. Once this has been adopted by internationally accepted Standards organizations such as ISO and BP it will make it harder and far more expensive for those who adulterate pure TTO to continue to dupe manufacturers and end-users for their own gain.
In the longer term, regulation is the other major challenge. Any natural substance varies slightly from year to year and from region to region but there are recognized standards in place that manage this so why can’t regulators also deal with the natural variation in these substances more sympathetically? On top of this there is also the prohibitive cost of developing data required in some jurisdictions to enable registration as a medicine or cosmetic ingredient. This can run into millions of dollars which cannot be recouped over the period a patent is usually granted for a conventional single molecule pharmaceutical because no patent is available for TTO. Traditional or customary use patterns covering many decades by bona fide practitioners should be acceptable evidence of the safety and efficacy of natural products such as TTO.
How does ATTIA help support the tea tree oil industry?
As an industry association, ATTIA provides both a forum and a vehicle for members (and non-members where necessary) to promote and act on emerging trends and regulatory, market or other pressures and trends in a timely and cohesive manner. By maintaining a monitoring watch on these factors ATTIA has been well placed to prepare and submit dossiers to regulatory bodies that include the USA, Canada and Europe. These submissions are prepared to ensure that pure Australian TTO can continue to be used in these jurisdictions both as a cosmetic ingredient and as a traditional herbal remedy for its wide range of clinically supported uses.
ATTIA also provides advice, training and auditing services in the critical area of quality assurance or QA. This promotion of ATTIA’s voluntary Code of Practice helps ensure that all pure Australian TTO offered into both international and local markets is always in perfect condition with documented QA practices that can be seamlessly incorporated into a manufacturer’s own QA procedures.
Since its inception ATTIA has also invested considerable time, effort and funding on research into the production, distillation, storage safety, efficacy and uses of pure Australian TTO. Much of this work, supported by ATTIA member donations and Australian Government Research and Development Grants, has been done at the University of Western Australia where the TTO Research Group has published more than 50 papers in their primary areas of research which are the antimicrobial and anticancer properties of TTO. Many of these articles are used to support claims and as proof of concept for the safety, efficacy and uses of pure Australian TTO. More on UWA here.